6 The Poisoning
For hundreds of millions of years, the mat communities thrived.
Picture Earth around 2.8 billion years ago.1 The atmosphere is a haze of nitrogen, carbon dioxide, methane, and water vapor. There is no ozone layer because there is nothing to make ozone from. Ultraviolet light hammers the surface unfiltered. The oceans are warm, slightly green from dissolved iron, and virtually without free oxygen – though trace amounts may have flickered locally around photosynthetic mats, the atmosphere held none.
In this world, the bacterial mats we met in the last chapter have built something remarkable: layered communities with stable energy cycles, closed chemical loops, a functioning economy. Sulfate reducers hand off waste to sulfur oxidizers. Methanogens scavenge hydrogen that fermenters discard. Electrons flow between guilds, waste is recycled, and the system is stable.
Then one lineage – the cyanobacteria – evolved a metabolism whose waste product was toxic to nearly every other organism on Earth.2
They learned to split water.
6.1 The world before the catastrophe
To understand why this was catastrophic, you have to understand what oxygen meant to early life. Not what it means to us – fuel for our mitochondria, the gas we can’t live five minutes without – but what it meant to them.
For most ancient forms of life on Earth, oxygen was a dangerous poison.3 It ripped electrons off proteins. It mangled DNA. It generated reactive molecules (what chemists now call “reactive oxygen species”) that shredded cell membranes.4 In a world that had evolved without it, there was no defense.
Even the cyanobacteria had no good defense against their own byproduct. They had invented the machinery to crack water molecules, but not the enzymes to neutralize the oxygen that resulted.
So why did they do it?
Because the prize was independence. Before oxygenic photosynthesis, every phototroph on Earth depended on a limited menu of electron donors – hydrogen sulfide, hydrogen gas, ferrous iron – to feed their light-harvesting machinery. These donors were not everywhere. They were concentrated near volcanic vents, hydrothermal systems, and specific chemical interfaces. A phototroph tethered to hydrogen sulfide was a phototroph tethered to geography.
Water, on the other hand, was everywhere.
The payoff was not primarily energetic but ecological: water was everywhere, offering a limitless supply of electrons to any organism that could crack it. The cost was a toxic byproduct. The result was ecologically devastating: cyanobacteria flooded the environment with a molecule that most other organisms had no defense against. But in the long run, oxygen opened an entirely new energy regime. Without them, Earth would still be a planet of microbes – and nothing more.
6.2 The ladder of electron donors
The transition from ancient photosynthesis to the water-splitting kind did not happen in a single leap. It happened in stages, and each stage was a revolution in its own right. To see why, we need to look at photosynthesis not as a single process but as a family of strategies, all built on the same chassis but plugged into different fuel lines.
The chassis is simple in concept: capture a photon of light, use its energy to boost an electron to a higher energy state, then pass that excited electron through a chain of proteins to do useful work. The question is: where does that electron come from in the first place?
In the earliest phototrophs, the electron donors were the easiest molecules to crack open. (The half-reactions below are written in standard acid form for electron bookkeeping; actual speciation varies with pH, and in-vivo coupling partners differ.)
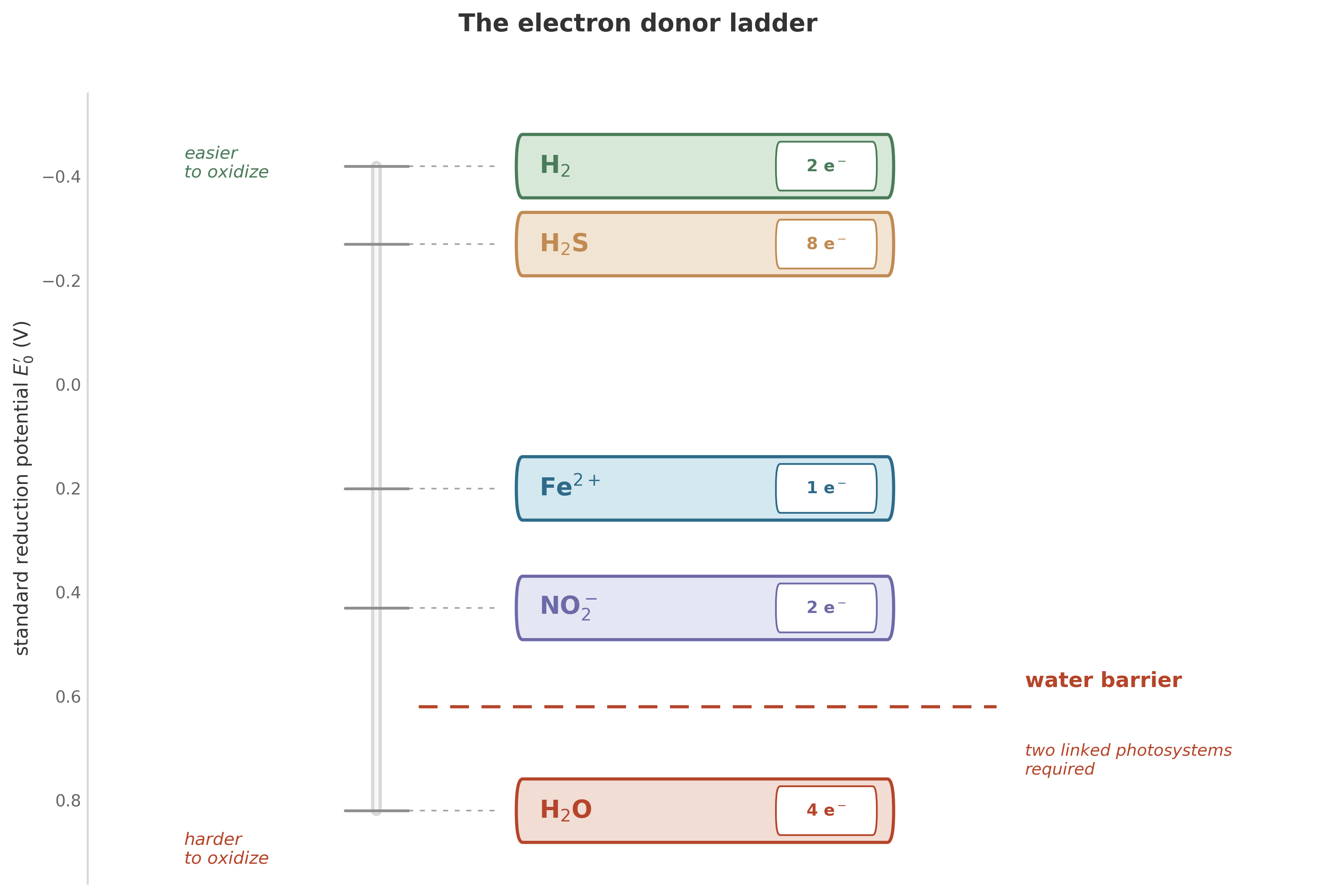
Hydrogen gas. The simplest donor. One molecule of H\(_2\) yields two protons and two electrons:
\[\text{H}_2 \longrightarrow 2\text{H}^+ + 2e^-\]
Hydrogen is easy to oxidize – the electrons practically fall off – but it is scarce in most environments. A metabolism built on hydrogen works beautifully near volcanic vents and poorly everywhere else.
Hydrogen sulfide. The workhorse of early photosynthesis. Sulfur bacteria – the purple and green sulfur clans – built vast communities around this donor:
\[\text{H}_2\text{S} + 4\,\text{H}_2\text{O} \longrightarrow \text{SO}_4^{2-} + 10\,\text{H}^+ + 8e^-\]
More electrons per molecule, and sulfide was abundant in the early oceans. But it was still a local resource, concentrated where volcanic gases met seawater.
Elemental sulfur. Some organisms could strip electrons from sulfur deposits directly:
\[\text{S} + 4\,\text{H}_2\text{O} \longrightarrow \text{SO}_4^{2-} + 8\,\text{H}^+ + 6e^-\]
Organic compounds. A few phototrophs could even oxidize simple organics like formaldehyde:
\[\text{CH}_2\text{O} + \text{H}_2\text{O} \longrightarrow \text{CO}_2 + 4\,\text{H}^+ + 4e^-\]
All of these share a common limitation. The electron donor is something the organism must find – a molecule delivered by geology, not biology. The phototroph is a tenant paying rent with whatever the environment provides. And in evolutionary terms, a tenant is always vulnerable to eviction.
6.2.1 An intermediate rung: nitrogen photosynthesis
In 1970, the biochemist John Olson published a theoretical model proposing that the transition from anoxygenic to oxygenic photosynthesis did not happen directly. There could have been an intermediate stage, he argued, in which organisms used nitrogen compounds – specifically nitrite – as electron donors.5
It was an elegant prediction. Nitrite sits between sulfide and water on the thermodynamic ladder of oxidation difficulty: harder to crack than sulfide, easier than water. If such organisms existed, they would represent the evolutionary equivalent of a smaller bridge before the big one.
But for thirty-seven years, no one could find the organism.
The prediction sat in the literature, cited occasionally, regarded as plausible but unconfirmed. Then, in 2007, at the University of Konstanz in Germany, a team of microbiologists ran a patient experiment. They grew photosynthetic bacteria under strictly anoxic conditions – no oxygen, no sulfide – and fed them nitrite (NO\(_2^-\)) as the only available electron donor.6
For weeks, nothing obvious happened. Then a faint pink color appeared in the culture. Pink is the telltale color of purple phototrophs – bacteriochlorophyll absorbing near-infrared light. And when they measured the chemistry, they found exactly what Olson had predicted: nitrite was being oxidized to nitrate (NO\(_3^-\)), and the energy was powering photosynthetic growth.
\[\text{NO}_2^- + \text{H}_2\text{O} \longrightarrow \text{NO}_3^- + 2\,\text{H}^+ + 2e^-\]
Thirty-seven years from prediction to confirmation. The organism had been there all along, doing what Olson said it should, in some quiet corner of the microbial world. It just needed someone to design the right experiment.
6.2.2 The final step: splitting water
These known donors can be arranged by oxidation difficulty, though whether evolution climbed them in exactly this order remains uncertain:
\[\text{H}_2 \rightarrow \text{H}_2\text{S} \rightarrow \text{N compounds} \rightarrow \text{H}_2\text{O}\]
Each step to the right means cracking a tougher molecule. Each step requires more sophisticated molecular machinery – a stronger oxidant at the heart of the photosystem. And each step liberates the organism from a scarcer resource and connects it to a more abundant one.
The final step – water – was the hardest and the most consequential:
\[2\text{H}_2\text{O} \longrightarrow 4\text{H}^+ + \text{O}_2 + 4e^-\]
Water is everywhere. An organism that can use water as its electron source is an organism that can photosynthesize anywhere there is light and water. It has no geographic constraint. It has no chemical dependency.7
It is also an organism that produces oxygen as waste.
6.3 How photosynthesis actually works (told briefly)
The molecular story is worth a paragraph, because it reveals something beautiful about the machinery – and something that will matter enormously when we get to respiration.
A quantum of light strikes a chlorophyll molecule. The photon’s energy kicks an electron in the chlorophyll to an excited state – a higher energy orbital, unstable and eager to fall. But instead of falling back and re-emitting the light (as a fluorescent dye would), the excited electron is caught by a neighboring protein and handed off to the next one in a chain. Each handoff drops the electron to a slightly lower energy level. Each drop releases a small packet of energy. That energy is used to pump protons across a membrane, building the electrochemical gradient that drives ATP synthesis.
At the end of the chain, the electron has lost most of its borrowed energy. It needs a home. In the simplest (cyclic) version of photosynthesis, the electron returns to the same chlorophyll molecule that launched it, and the cycle repeats. In the more elaborate version – the one cyanobacteria invented – two photosystems work in series, and the electron ends up reducing CO\(_2\) to organic carbon. The carbon in CO\(_2\) starts at an oxidation state of +4; by the time it is incorporated into sugar, it has been reduced. The electron has done real chemical work.
But there is a catch. If the electron departs chlorophyll and travels down the chain to reduce carbon, it does not come back. The chlorophyll is left with a hole – an electron vacancy. Something must refill it.
In anoxygenic phototrophs, the refill comes from sulfide, or hydrogen, or one of the other donors we listed. In cyanobacteria, the refill comes from water. And when you rip an electron from water, the leftovers are protons and molecular oxygen.
Viruses now sit inside this machinery too. About four out of five sequenced cyanophage genomes carry psbA, the gene for the D1 core protein of Photosystem II, and many carry psbD as well.8 A phage that knocks out photosynthesis too soon cuts its own replication short, so it carries the parts that keep the host’s light reactions running during infection. Some researchers argue that this long gene traffic between cyanobacteria and their phages may have helped shape the early history of oxygenic photosynthesis. The evidence does not make viruses the prime mover. It does make them part of the story.
That is the entire invention. The rest is consequences.
6.4 The Great Oxidation Event
The consequences took time to arrive. Cyanobacteria probably evolved oxygenic photosynthesis somewhere between 2.7 and 3.0 billion years ago – the exact date is debated, and the geological evidence remains ambiguous.9 But for hundreds of millions of years after the invention, free oxygen did not accumulate in the atmosphere. It was consumed as fast as it was produced, mopped up by a planet full of reduced minerals and dissolved iron that reacted with oxygen instantly.10
Think of it as a bathtub with the drain open. The faucet is running – cyanobacteria are producing oxygen – but the drain is bigger. Reduced iron in the oceans, sulfide in volcanic gases, reduced minerals on land: all of these were oxygen sinks, chemical sponges that soaked up every molecule of O\(_2\) before it could accumulate.
Slowly, over hundreds of millions of years, the sinks filled. The reduced iron precipitated out as iron oxides – the banded iron formations that today form some of the world’s richest ore deposits.11 The sulfide was oxidized. The easily reacted minerals were used up. The bathtub drain narrowed.
And then, around 2.4 billion years ago, the faucet won.12
Oxygen began to accumulate in the atmosphere. Not much by modern standards – perhaps 1 to 2 percent of present levels at first – but enough to reshape the chemistry of the planet’s surface.13 This is the Great Oxidation Event, and it was, in the precise language of geochemistry, a catastrophe.
But the phrase “the Great Oxidation Event” is slightly misleading if it sounds too singular. The rise of oxygen was not a clean flip from off to on. Geochemical proxies preserve hints of earlier oxygen “whiffs,” and the interval after the main rise appears to have included repeated oscillations between more oxic and more reducing states before the atmosphere settled into a new regime.1415 The surface ocean, the deep ocean, and the atmosphere did not oxygenate in lockstep. The poisoning was staggered, uneven, and regionally messy – which is exactly what you would expect from a planet whose sinks, sources, and microbial communities were all changing together.
For the anaerobic communities that had built the living world, free oxygen was lethal. Organisms that had never encountered this molecule – had never needed defenses against it – suddenly found their enzymes damaged, their membranes compromised, their DNA under attack. What may have been Earth’s first mass extinction was not caused by an asteroid or a volcanic eruption. It was caused by a microbe’s waste product.
The survivors retreated. They found refuge in the places oxygen could not reach: deep sediments, waterlogged soils, the interiors of organic-rich deposits, the guts of other organisms. Many of the anaerobic metabolisms we study today – sulfate reduction, methanogenesis, iron reduction – are practiced by lineages whose ancestors were driven underground by the Great Oxidation Event. They are survivors of the poisoning, confined to anoxic refugia for 2.4 billion years.
The poisoning itself took a few hundred million years. Its consequences are still unfolding. What follows is not the full story – that would take several chapters – but a tour of the most important aftershocks: how life learned to breathe the poison, how it coped with the chemical incompatibilities oxygen created, how captured cyanobacteria became the engines of every plant, and how the energy surplus oxygen provided underwrote the complexity of everything that followed.
6.5 The invention of respiration
The same molecular machinery that made oxygen deadly also made oxygen useful. And the organisms that figured this out first were likely cyanobacteria or their close relatives – though the evolutionary pathway was probably less clean than a single invention event, with multiple lineages independently recruiting components of the electron transport chain.
The logic is startlingly simple once you see it. Photosynthesis works by passing excited electrons down a chain of protein complexes, harvesting energy at each step. The electron starts at chlorophyll (boosted by light) and ends at a carbon compound (reducing CO\(_2\)).
Now imagine a small modification. Instead of starting the electron at chlorophyll, start it at an organic molecule – say, pyruvate, a common product of fermentation. And instead of ending the chain at CO\(_2\), end it at oxygen. The electron still passes through the same protein complexes. The energy is still harvested in the same way – proton pumping, ATP synthesis, the whole apparatus. But now the process runs in reverse conceptual direction: instead of using light to push electrons uphill and fix carbon, it lets electrons roll downhill from organic carbon to oxygen, and captures the energy released.16
Aerobic respiration comes from that reversal. It solved two problems at once: oxygen received electrons and turned into water, while the cell captured a large energy gain in the process.17
The deep irony is that respiration is carried out by the same protein complexes as photosynthesis. The cytochrome chains, the proton-pumping machinery, the ATP synthase – all of it is shared, or at least derived from the same ancestral toolkit. In modern cyanobacteria, photosynthesis and respiration use overlapping components to such an extent that there is a kind of competition between the two processes for the right to use the same proteins.18
It is a record of evolutionary history. Respiration was not invented from scratch. It was photosynthesis repurposed – the machinery of light-harvesting retooled to run on chemical fuel, with oxygen as the terminal electron acceptor instead of chlorophyll as the starting electron donor.
Purple bacteria, close relatives of the cyanobacteria, found the same solution with what some researchers argue was even greater efficiency. The molecular details differ, but the principle is identical: use the existing electron transport chain, swap the electron source and sink, and suddenly you have a metabolism that thrives on the very poison that was killing everything else.
When comparing different metabolisms – iron reduction versus sulfate reduction, say, or aerobic versus anaerobic respiration – the temptation is to compare energy yields per mole of reaction. This can be misleading, because different reactions transfer different numbers of electrons. The fair comparison is energy yield per electron transferred.19
This matters practically because microbial rate laws in reactive transport models need to respect thermodynamics. A reaction that is thermodynamically favorable on paper may stall if local concentrations push it close to equilibrium. The thermodynamic factor \(F_T\) captures this:
\[F_T = \frac{1}{\exp\!\left(\frac{\Delta G_r}{RT}\right) + 1}\]
where \(\Delta G_r\) is the in-situ Gibbs energy of the reaction.202122
Read \(F_T\) as a smooth switch. Far from equilibrium (\(\Delta G_r\) is large and negative), \(F_T \approx 1\) and the reaction proceeds at its full kinetic rate. In this simplified form, \(F_T\) reaches \(1/2\) at equilibrium (\(\Delta G_r = 0\)). The full Jin-Bethke treatment adds a term for the energy the cell conserves per reaction (ATP synthesis), which shifts the effective cutoff into negative \(\Delta G_r\) territory – so that in practice, the reaction throttles to near zero well before thermodynamic equilibrium. There is no sharp cutoff, just a gradual throttle, which is exactly how real microbial communities behave. See Chapter 8 for the full treatment.
Temperature also matters. The Arrhenius relation gives the temperature dependence of the rate constant:
\[k(T) = A\,\exp\!\left(-\frac{E_a}{RT}\right)\]
where \(E_a\) is the apparent activation energy.23 A caveat: the Arrhenius equation was derived for elementary reactions. When applied to complex microbial processes, the fitted \(E_a\) values are “apparent” – empirical summaries of many underlying steps. Arrhenius strictly relates the rate constant \(k\) to temperature, not the rate itself; the rate also depends on substrate concentrations, biomass, and other factors.24
6.6 The day shift and the night shift
Oxygen created another problem, one that reveals the precision of microbial adaptation.
Nitrogen fixation – the conversion of atmospheric N\(_2\) into biologically usable ammonia – is one of the most important reactions in the biosphere. Without it, life would be perpetually starved for nitrogen, the element needed for every protein and every nucleic acid. The enzyme that performs this reaction, nitrogenase, is ancient, elaborate, and spectacularly sensitive. It cannot work in the presence of oxygen.25
For anaerobic organisms, this was never a problem. But for cyanobacteria – organisms that produce oxygen as a byproduct of their core metabolism – it was a biochemical incompatibility. How do you fix nitrogen when your own photosynthesis is flooding the cell with the one substance that destroys the nitrogen-fixing enzyme?
Many cyanobacteria solved this with specialized cells called heterocysts: thick-walled compartments that exclude oxygen and dedicate themselves entirely to nitrogen fixation while neighboring cells handle photosynthesis – a spatial solution to a chemical incompatibility.26
But some cyanobacteria had a more elegant solution, and it took until 2006 to discover it.
In the hot springs of Yellowstone National Park, microbial mats dominated by the cyanobacterium Synechococcus had long puzzled researchers. These mats thrive at temperatures above 50 degrees Celsius – too hot for the filamentous cyanobacteria that typically fix nitrogen in mat communities. And yet the mats clearly were not nitrogen-starved. Where was the nitrogen coming from?
The answer turned out to be time.27
Synechococcus runs photosynthesis during the day and nitrogen fixation at night. During daylight hours, the cells photosynthesize normally, producing oxygen and fixing carbon. As the sun sets, photosynthesis slows, then stops. Oxygen concentrations in the mat plummet – consumed by respiration and no longer replenished by photosynthesis. And in the darkness, with oxygen safely absent, the cells switch on their nitrogenase genes and begin fixing nitrogen.
By dawn, they switch back.
The switch is graded, not binary. The researchers found a tightly choreographed sequence of gene expression: photosynthesis genes ramping down in the evening, fermentation genes ramping up (to provide the ATP needed for nitrogen fixation in the absence of light), nitrogenase genes peaking in the dark hours, and then the whole program reversing at sunrise. A single cell, running two incompatible metabolisms, separated not by walls but by the clock.
The discovery explained something that had bothered microbiologists for years. Scientists had believed that combining photosynthesis and nitrogen fixation in the same cell – without heterocysts – was impossible. Synechococcus proved that impossibility is sometimes just a failure of imagination. Time is a compartment too.
6.7 Houses for cyanobacteria
There is a line, sometimes repeated among biologists with a taste for provocation, that goes like this: plants are just comfortable houses built for the convenience of cyanobacteria.28
It sounds like a joke. It is not.
Every plant cell that photosynthesizes does so using organelles called chloroplasts. Chloroplasts have their own DNA. They have their own ribosomes. They divide independently of the host cell. Their genome is unmistakably cyanobacterial – not “similar to” cyanobacteria in the way that a cousin resembles a cousin, but derived from cyanobacteria in the way that a captured soldier becomes a citizen of the conquering nation.
Sometime between 1.0 and 1.5 billion years ago, a eukaryotic cell engulfed a cyanobacterium and did not digest it.29 Instead, over vast stretches of time, the two organisms merged. The cyanobacterium lost most of its genes to the host’s nucleus – stripped of its independence, reduced to an organelle. But it kept the one thing that mattered: the photosynthetic machinery. The water-splitting, oxygen-producing, carbon-fixing apparatus that cyanobacteria had invented a billion years earlier.
Every leaf on every tree, every blade of grass, every strand of kelp in the ocean – all of them photosynthesize using the machinery that cyanobacteria invented more than two billion years ago, housed inside eukaryotic cells that provide structure, protection, and nutrient supply.
The cyanobacteria poisoned the world. Then they moved indoors.
6.8 The nitrogen bargain
The poisoning had one more consequence worth telling, because it created one of the most important symbioses in the living world – and one that still shapes human civilization.
Plants need nitrogen. They need it for amino acids, for nucleotides, for chlorophyll itself. But most plants cannot fix nitrogen. The triple bond in N\(_2\) is one of the strongest in chemistry,30 and nitrogenase – the only enzyme that can break it – belongs to bacteria, not to plants. In most terrestrial ecosystems, the lack of available nitrogen is the main factor limiting plant growth. Remove that limitation and productivity explodes.31
Evolution’s answer was partnership. Across many lineages, plants entered into symbioses with nitrogen-fixing bacteria: cyanobacteria in some cases, actinobacteria in others, and most famously, alpha-proteobacteria of the genus Rhizobium in the legumes.32 The arrangement is always the same in principle. The plant builds a specialized structure (a root nodule, a leaf cavity, a thickened stem) that provides a low-oxygen environment – because nitrogenase still cannot tolerate oxygen, even 2.4 billion years after the poisoning. The bacterium moves in, fixes nitrogen, and shares ammonia with the host. In return, the plant feeds the bacterium sugars produced by photosynthesis.
Agriculture runs on that deal. Every soybean field, every clover pasture, every acacia tree in the savanna depends on a partnership between a plant and a bacterium that can fix nitrogen in a world full of the gas that destroys the enzyme needed to do it. The whole arrangement works around the consequences of the Great Oxidation Event. The root nodule shelters nitrogenase from a poison released 2.4 billion years ago.
6.9 The energetics of a new world
The Great Oxidation Event did not merely change the atmosphere. It rewrote the energy budget of the biosphere.
Consider the numbers. Anaerobic metabolisms – fermentation, sulfate reduction, methanogenesis – extract energy from organic molecules, but they leave much of the potential energy locked in the products. Fermentation of glucose to ethanol, for instance, captures only a fraction of the total energy available in the glucose molecule. The ethanol still has electrons to give; the organism simply cannot access them without a more powerful electron acceptor.
Oxygen changes this calculation entirely. As a terminal electron acceptor, oxygen sits at the bottom of the thermodynamic hill – one of the strongest oxidants in the biological world. An organism that can pass electrons all the way from organic carbon to oxygen extracts far more energy per molecule of food than any anaerobic metabolism can. Where fermentation yields about 2 ATP per glucose, aerobic respiration yields roughly 30–32 – an increase of more than an order of magnitude, though the exact ratio varies with organism and pathway.33
The difference is large. Anaerobic organisms survive; aerobic organisms thrive. They grow faster, maintain larger cells, and can afford the energetic overhead of complex internal structures. The rise of large, complex eukaryotic cells – and later multicellular life – tracks that shift. Oxygen made far more energy available per molecule of food, and complex life spent it.
The irony is inescapable. The greatest environmental catastrophe in Earth’s history – a mass poisoning that drove most of the biosphere into hiding – was also the event that made complex life possible. Without oxygenic photosynthesis, there would be no animals, no fungi, no plants. The bacterial mat communities would still be the pinnacle of biological organization, cycling sulfur and methane in their closed loops, stable and productive and unable to build anything larger than a film of slime.
6.10 The long aftermath
The transition was not clean. The Great Oxidation Event was followed by hundreds of millions of years of fluctuation – periods when oxygen rose, crashed, and rose again. The “Boring Billion,” as some geologists call the period from roughly 1.8 to 0.8 billion years ago, saw oxygen levels stabilize at a fraction of modern values – enough to sustain aerobic life in surface waters, not enough to oxygenate the deep ocean.34 The deep waters remained anoxic, or at best “ferruginous” (iron-rich and oxygen-free), for most of this interval.
Researchers now treat the delay as a systems problem, not a single missing switch. Oxygenic photosynthesis may have originated hundreds of millions of years before atmospheric oxygen finally broke through, with photochemical destruction, nutrient bottlenecks, and ocean chemistry all suppressing the signal.35 A recent proposal adds another constraint: high nickel and urea concentrations in Archean oceans may have held cyanobacterial productivity below the threshold needed to oxygenate the atmosphere at scale.36 Cyanobacteria did not invent oxygen one day and transform the planet the next. Geology, ocean chemistry, and nutrient supply kept pushing back.
This means that for over a billion years, Earth was chemically partitioned. The surface was a new world – oxygenated, dangerous to anaerobes, open to aerobic innovation. The deep ocean and the sediments remained an old world – anoxic, sulfidic or ferruginous, still running on the ancient metabolisms. The two worlds coexisted, separated by a chemical boundary that shifted with the seasons, the currents, and the slow rhythms of plate tectonics.
You can still see that boundary. In every stratified lake, every oxygen-minimum zone in the ocean, every waterlogged soil, there is a depth where oxygen disappears and the old metabolisms take over. The sulfate reducers, the methanogens, the iron reducers – they never left. Oxygen pushed them to the margins.
Modern Earth is not an oxygen planet that happens to contain some anaerobic pockets. It is a planet where two worlds – one oxidized, one reduced – have coexisted since the Great Oxidation Event, separated by gradients that microbes both create and exploit. The porewater profiles that geochemists measure in sediments today are the living record of that 2.4-billion-year-old partition.
6.11 The ledger
The cyanobacteria cracked water, liberating electrons that had been locked in the most abundant molecule on the planet’s surface. In doing so, they produced a toxic waste product that drove the majority of existing life into exile.
The same molecular machinery that produced the poison was then repurposed – by the cyanobacteria themselves, or by their neighbors – into a mechanism for consuming it. Respiration turned the poison into the most efficient electron acceptor biology had ever used.
The enzyme that fixes nitrogen – ancient, essential, irreplaceable – could not tolerate the new atmosphere. So organisms compartmentalized: some in space (heterocysts), some in time (the day-night switch of Synechococcus). Later, plants built shelters for nitrogen-fixing bacteria in their roots, recreating pockets of the pre-oxygen world inside their own tissues.
The cyanobacteria themselves were eventually captured, domesticated, and converted into the chloroplasts of every photosynthetic eukaryote on Earth. Their descendants now live inside plant cells, still splitting water, still producing oxygen, still running the same ancient machinery – but housed, fed, and protected by the organisms that evolved in the world they created.
And the energy surplus that oxygen provided – more than an order of magnitude more ATP per molecule of food – made possible the construction of large, complex cells, and eventually large, complex organisms. Every animal alive today runs on aerobic respiration. Every breath you take is a transaction with cyanobacterial legacy.
The Great Oxidation Event killed much of what lived and confined the survivors to anoxic refugia. Over billions of years, the new redox landscape enabled cells to extract far more energy from their food – enough to build complexity.
The molecule that was once the deadliest waste product became the most efficient electron acceptor in biology. And the cyanobacteria that produced it became, as chloroplasts, the photosynthetic engines inside every plant cell on Earth.
The Archean atmosphere (3.8-2.5 Ga) was reducing, dominated by N\(_2\), CO\(_2\), CH\(_4\), and H\(_2\)O vapor, with negligible free oxygen. The absence of ozone allowed intense UV radiation to reach the surface. See Kasting (1993) for atmospheric evolution models. (Kasting 1993)↩︎
Cyanobacteria evolved oxygenic photosynthesis between 2.7 and 3.0 Ga, though the exact timing remains debated. Buick (2008) reviews the geological and geochemical evidence for the origin of oxygenic photosynthesis. (Buick 2008)↩︎
Molecular oxygen generates reactive oxygen species (superoxide, hydrogen peroxide) that damage proteins, DNA, and cell membranes. For a comprehensive review of oxidative damage pathways, see Imlay (2003). (Imlay 2003)↩︎
Reactive oxygen species (ROS) include superoxide (O\(_2^-\)), hydrogen peroxide (H\(_2\)O\(_2\)), and hydroxyl radicals (OH\(\cdot\)). These molecules oxidize iron-sulfur clusters in proteins, abstract hydrogen atoms from lipids, and damage nucleic acids. The chemistry of ROS in biological systems is reviewed in Imlay (2003). (Imlay 2003)↩︎
J. M. Olson, “The Evolution of Photosynthesis,” Science 168 (1970): 438–446. Olson proposed that nitrogen compounds served as intermediate electron donors in the evolutionary transition from anoxygenic to oxygenic photosynthesis. (Olson 1970)↩︎
B. M. Griffin, J. Schott, and B. Schink, “Nitrite, an Electron Donor for Anoxygenic Photosynthesis,” Science 316 (2007): 1870. The first demonstration that anoxygenic phototrophs can use nitrite as their sole electron donor, confirming Olson’s 37-year-old prediction. (Griffin et al. 2007)↩︎
The evolution of oxygenic photosynthesis required linking two photosystems in series to generate sufficient reduction potential to split water. Blankenship (2010) provides a comprehensive review of photosynthetic evolution. (Blankenship 2010)↩︎
Iwona Slesak and Halina Slesak, “Cyanophages as an important factor in the early evolution of oxygenic photosynthesis,” Scientific Reports 12 (2022). The paper surveys cyanophage
psbAandpsbDgenes and argues that phage-host gene traffic belongs in the longer history of Photosystem II. (Slesak and Slesak 2022)↩︎Molecular clock estimates and biomarker evidence suggest cyanobacteria evolved between 2.7 and 3.0 Ga, but free oxygen did not accumulate until ~2.4 Ga. The timing and environmental context are reviewed in Buick (2008) and Lyons et al. (2014). (Buick 2008; Lyons et al. 2014)↩︎
Before atmospheric oxygen accumulation, O\(_2\) produced by cyanobacteria was consumed by reduced minerals (particularly dissolved Fe\(^{2+}\)), volcanic gases (H\(_2\), H\(_2\)S), and organic matter. Holland (2006) quantifies these oxygen sinks. (Holland 2006)↩︎
Banded iron formations (BIFs) are sedimentary deposits consisting of alternating layers of iron oxides and silica, formed when dissolved Fe\(^{2+}\) was oxidized and precipitated. BIFs peaked around 2.5 Ga and represent a major oxygen sink. Klein (2005) reviews BIF formation and distribution. (Klein 2005)↩︎
The Great Oxidation Event occurred ~2.4 Ga, marked by the disappearance of mass-independent sulfur isotope fractionation and the appearance of red beds (oxidized continental sediments). Holland (2006) and Lyons et al. (2014) provide detailed reviews. (Holland 2006; Lyons et al. 2014)↩︎
Post-GOE atmospheric oxygen levels were initially 1-10% of present atmospheric level (PAL), far below modern 21%. Oxygen rose in stages, reaching near-modern levels only in the late Neoproterozoic. See Lyons et al. (2014) and Catling and Claire (2005). (Lyons et al. 2014; Catling and Claire 2005)↩︎
Ariel D. Anbar et al., “A whiff of oxygen before the Great Oxidation Event?” Science 317 (2007): 1903–1906. Redox-sensitive metal enrichments in the Mount McRae Shale suggest transient oxygenation before permanent atmospheric oxygen buildup. (Anbar et al. 2007)↩︎
Dilan M. Ratnayake et al., “Biogeochemical impact of nickel and urea in the Great Oxidation Event,” Communications Earth & Environment (2025). The study proposes that high nickel and urea concentrations in Archean oceans may have helped delay cyanobacterial expansion and therefore delayed oxygenation. (Ratnayake et al. 2025)↩︎
Aerobic respiration likely evolved in bacteria closely related to cyanobacteria, repurposing components of the photosynthetic electron transport chain. Lane (2005) discusses the energetic advantages and evolutionary origins of aerobic respiration. (Lane 2005)↩︎
Castresana and Moreira (1999) demonstrated that respiratory chains share deep evolutionary ancestry with photosynthetic electron transport, supporting the view that aerobic respiration was repurposed from the photosynthetic machinery. (Castresana and Moreira 1999)↩︎
In modern cyanobacteria, the respiratory and photosynthetic electron transport chains share several protein complexes, including the cytochrome b\(_6\)f complex and the plastoquinone pool. For a review, see Blankenship (2010). (Blankenship 2010)↩︎
LaRowe and Van Cappellen (2011) show that comparing catabolic yields on a per-electron basis provides a thermodynamically consistent framework for understanding microbial energetics across diverse metabolisms. (LaRowe and Van Cappellen 2011)↩︎
Jin and Bethke (2005) developed thermodynamic rate laws for microbial respiration that smoothly transition from kinetic to thermodynamic control as reactions approach equilibrium. (Jin and Bethke 2005)↩︎
Dale et al. (2006) applied thermodynamic inhibition functions to model organic matter degradation in marine sediments, showing how near-equilibrium conditions slow microbial rates. (Dale et al. 2006)↩︎
Regnier et al. (2011) provide a comprehensive review of biogeochemical reaction networks in aquatic sediments, emphasizing the role of thermodynamic controls on microbial metabolism. (Regnier et al. 2011)↩︎
Middelburg et al. (1996) compiled apparent activation energies for organic matter mineralization in marine sediments, finding values typically between 40-100 kJ/mol. (Middelburg et al. 1996)↩︎
Sandra Arndt et al., “Quantifying the Degradation of Organic Matter in Marine Sediments: A Review and Synthesis,” Earth-Science Reviews 123 (2013): 53–86. Apparent activation energies in diagenetic models aggregate multiple temperature-sensitive processes. (Arndt et al. 2013)↩︎
The nitrogenase enzyme, responsible for biological nitrogen fixation, is irreversibly inactivated by molecular oxygen. For structural and mechanistic details, see Howard and Rees (1996). (Howard and Rees 1996)↩︎
Heterocysts are specialized cells in filamentous cyanobacteria that provide an anoxic microenvironment for nitrogen fixation. They have thickened cell walls to limit oxygen diffusion and lack photosystem II. Adams (2000) reviews heterocyst structure and function. (Adams 2000)↩︎
A.-S. Steunou et al., “In situ analysis of nitrogen fixation and metabolic switching in unicellular thermophilic cyanobacteria inhabiting hot spring microbial mats,” PNAS 103 (2006): 2398–2403. (Steunou et al. 2006)↩︎
“Some biologists say, using metaphorical language, that plants are just comfortable houses for the living of cyanobacteria.” Markov, Birth of Complexity. (Markov 2010)↩︎
Chloroplasts originated from the endosymbiosis of a cyanobacterium by a eukaryotic host ~1.5 Ga. Keeling (2010) reviews the molecular and genomic evidence for plastid origins. (Keeling 2010)↩︎
The N≡N triple bond has a bond dissociation energy of 945 kJ/mol, one of the strongest in chemistry. Breaking this bond requires the complex nitrogenase enzyme system. Howard and Rees (1996) describe the structural basis of nitrogen fixation. (Howard and Rees 1996)↩︎
Nitrogen limits net primary production in most terrestrial biomes and many marine ecosystems. Vitousek and Howarth (1991) provide a comprehensive analysis of why nitrogen limitation is so pervasive. (Vitousek and Howarth 1991)↩︎
Legumes form root nodules housing nitrogen-fixing rhizobia. The plant provides sugars and maintains low oxygen concentrations; the bacteria fix N\(_2\) and supply ammonia. This symbiosis is reviewed in Margulis (1998) and Knoll (2003). (Margulis 1998; Knoll 2003)↩︎
Aerobic respiration of one glucose molecule yields ~30-32 ATP via glycolysis, the citric acid cycle, and oxidative phosphorylation. Fermentation yields only 2 ATP per glucose. The energetic basis is discussed in Lane (2005). (Lane 2005)↩︎
The Mesoproterozoic “Boring Billion” (1.8-0.8 Ga) was characterized by low, stable atmospheric oxygen levels, muted tectonic activity, and limited biological innovation. Holland (2006) and Lyons et al. (2014) discuss this interval. (Holland 2006; Lyons et al. 2014)↩︎
Julia Elizabeth Horne, Colin Goldblatt, and Lee Kump, “An early origin of oxygenic photosynthesis delays the Great Oxidation,” Philosophical Transactions of the Royal Society B (2025). The EONS model shows that an early origin of oxygenic photosynthesis is compatible with a delayed GOE once nutrient and redox feedbacks are included. (Horne et al. 2025)↩︎
Dilan M. Ratnayake et al., “Biogeochemical impact of nickel and urea in the Great Oxidation Event,” Communications Earth & Environment (2025). The study proposes that high nickel and urea concentrations in Archean oceans may have helped delay cyanobacterial expansion and therefore delayed oxygenation. (Ratnayake et al. 2025)↩︎